 
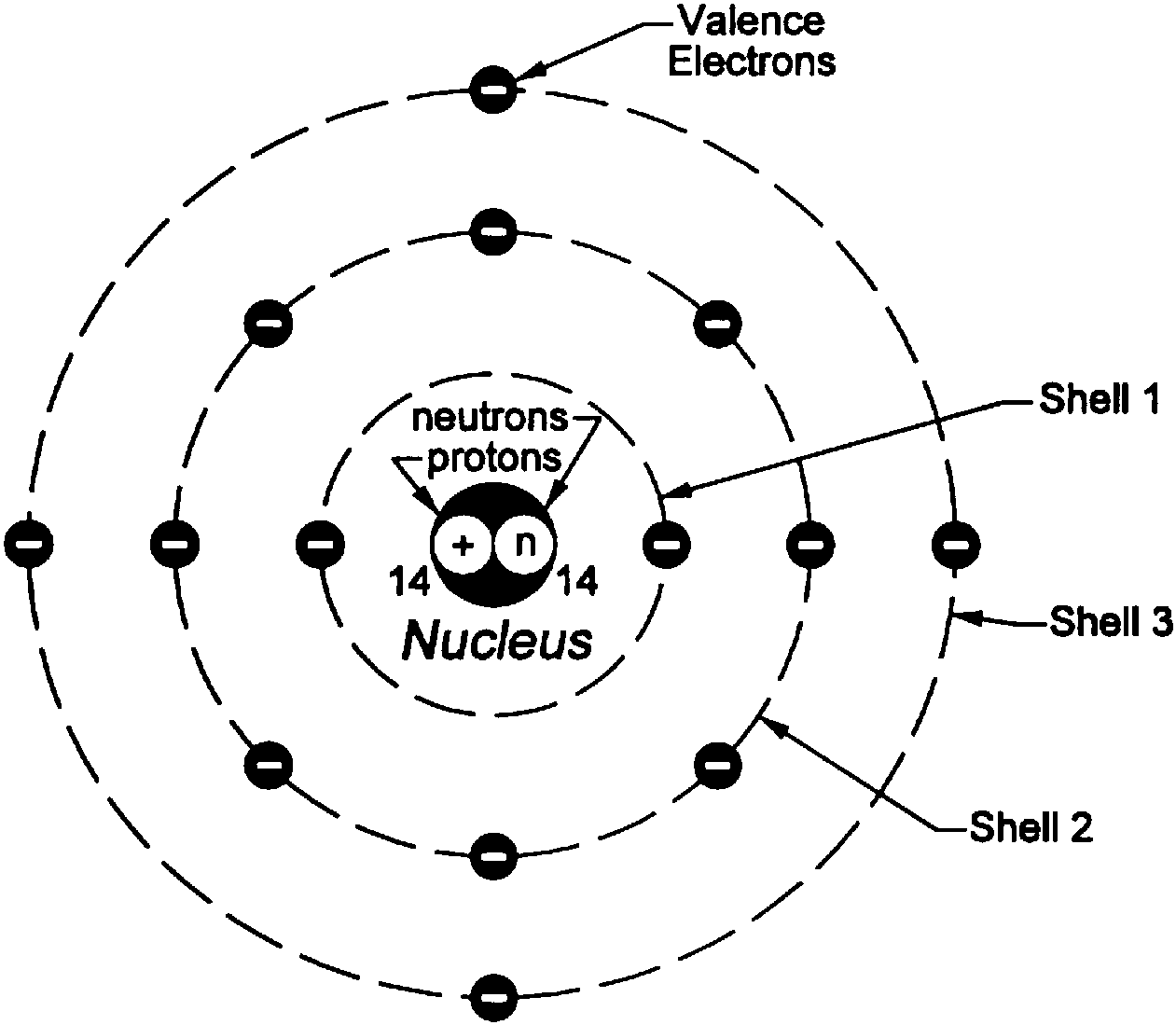
(starting from the bottom, closest to the nucleus, electrons could be at n 1, 2, 3, etc.) As they get 'excited', they move up. With orbitals (the Quantum Mechanical Model), electrons change positions With the Bohr Model, electrons are at a fixed position. Bohr model is utilized for giving a lucid explanation to the features of an atom. With orbits (the Bohr Model) elections are at a fixed positions. 
"Sommerfeld formula and Dirac's theory" (PDF). Niels Bohr proposed a new atomic model by giving a better explanation to the Rutherford model. In that model, the positive charges made up the pudding, while the electrons acted as. 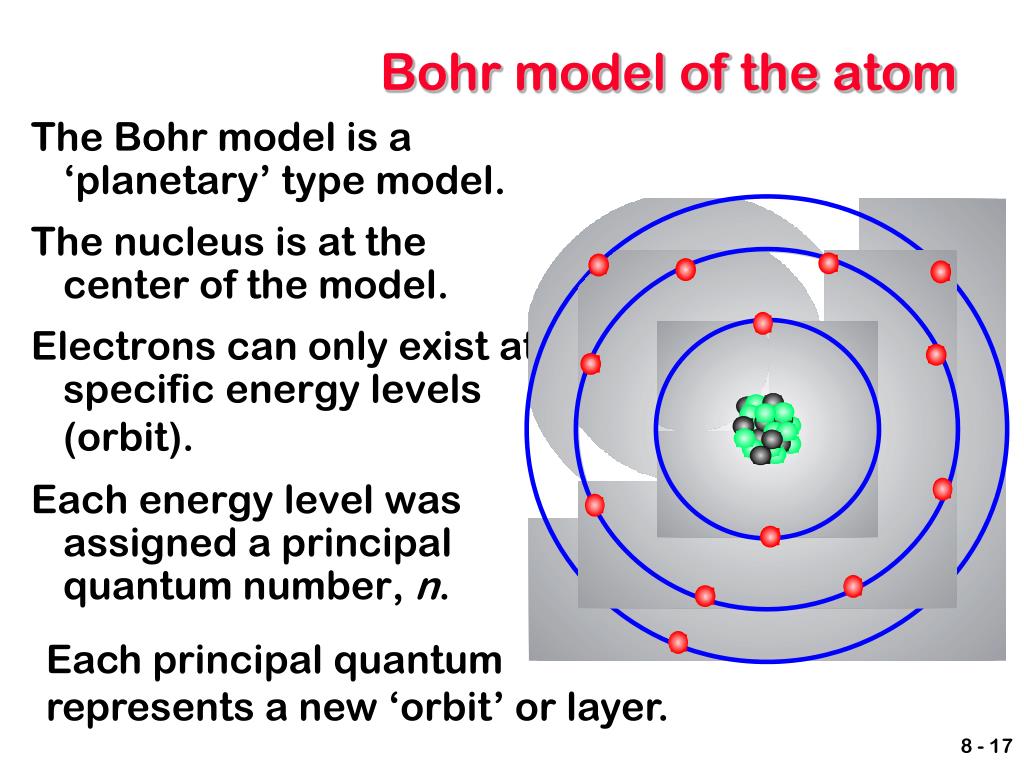
Thomson, the model suggested that the atom was a spherical ball of positive charge, with negatively charged electrons scattered evenly throughout.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
